The Military Medical Academy of Vietnam is looking for 20 healthy volunteers who can participate in the first clinical tests of a Vietnamese-made COVID-19 vaccine, with tests due to start on December 10.
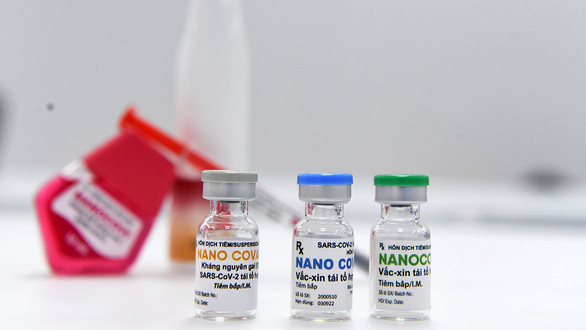 |
|
Vietnamese-made COVID-19 vaccine (Source: tuoitre.vn)
|
A small group of up to two volunteers are set to be initially vaccinated, and they will be monitored for reactions over the subsequent 24 hours to 72 hours, before going ahead with injections on the remaining volunteers, reported VOV.
Following three months of trials on the first group of volunteers, the COVID-19 vaccine will subsequently be tested on approximately 400 people.
The Military Medical Academy has stated that it has completed all relevant logistical preparations ahead of the clinical trials, including solutions for dealing with undesirable reactions that may occur following the injection.
A representative from the Ministry of Health indicates that the Ethics Council of the Ministry will carefully consider necessary steps to take with regard to human vaccine testing.
According to experts, those who are selected for participation in the COVID-19 vaccine trials must be healthy volunteers who are suffering from no underlying illnesses and have no medical history. These individuals will be carefully questioned about their health history, along with any allergies to drugs, food, or other things before they get vaccinated.
Similarly to drugs, vaccines may cause undesirable reactions, meaning that it is important for each phase of the vaccination process to be closely monitored by specialists in order to ensure the vaccine’s safety and efficiency.
A health expert believes that the initial assessments of COVID-19 vaccines globally are comparatively safe. Indeed, locally-produced COVID-19 vaccines have been proven to be safe on animals, although the clinical trials conducted on humans require strict conditions to ensure their safety and efficiency.
A total of three Vietnamese vaccine producers, Ivac, Vabiotech, and Nanogen, have completed the relevant COVID-19 vaccine production process at a laboratory scale and are now evaluating the vaccines’ safety and immunity on animals.
Nanogen and the Military Medical Academy are recruiting volunteers for the first phase of clinical trials which will run on December 10.
BTA